One of our most effective cancer treatments works like strategies developed to catch robbers. As they speed away in their get-away car from a heist, police throw spike-traps (Figure 1) in front of robbers to pop their tires, thwarting their escape. Throwing caution to the wind and driving at break-neck speeds was supposed to get the thieves away, but just led to their capture. A defining feature of cancer cells is their rapid, unchecked growth. We take advantage of this unique feature in radiation therapy, one of the first, and still most commonly employed cancer treatments. Radiation causes DNA damage that preferentially kills cancer cells because they speed past important quality-control steps in favor of growing rapidly. It is targeted to the location of tumors, further sparing healthy cells.

Figure 1. Spike-trap1.
When robbers go to look at their spoils, a dye bomb hidden inside stacks of cash can explode and mark them for easy detection. Radiation has another function as well: just like dye bombs, radiation makes cancer cells more apparent to our immune system and facilitates detection. Here, we will discuss what radiation is, what it does to cells, and how it specifically eliminates cancer cells.
What is radiation?
Before we talk about how it kills cells, let’s discuss radiation (just a bit – don’t worry, this is not a physics-focused article). Molecules that make up our cells consist of a series of atoms, the fundamental building block of life. Atoms are formed by a core of particles called protons and neutrons, surrounded by electrons. The atoms that make up our cells, mostly carbon, hydrogen, nitrogen, and oxygen, are connected by chemical bonds. Chemical bonds are a sort of “atomic glue”. These bonds are strong but can be broken with enough energy, from sources like heat and radiation. Radiation naturally comes from unstable atoms. Some atoms, like uranium and plutonium, are considered “unstable,” meaning they have an unbalanced number of neutrons and protons, causing the atom to have excess energy. To achieve stability, these atoms spontaneously shoot out particles in the form of radiation2.
How radiation causes DNA damage
For therapy, the first approaches, that are occasionally still employed today, harnessed more controllable unstable elements such as cobalt or iodine. Recently, most radiation therapy employs multiple particle types produced artificially with machines. DNA damage is caused directly by radiation, but also indirectly: indiscriminate damage to chemical bonds breaks water molecules, producing reactive oxygen species (ROS), like hydrogen peroxide (H2O2), capable of causing DNA damage (Figure 2)3.
A defining feature of radiation is its ability to pass through objects. At very low doses, radiation causes minimal chemical bond damage, but passes through different materials at different rates, allowing us to see inside of them – think X-rays used for broken bones, or to see inside of our baggage at airport security checkpoints. At higher doses, radiation will both pass through materials, and cause DNA damage. Because X-rays can pass through our bodies, healthy cells are often collateral damage during cancer treatment. We mitigate their exposure by limiting radiation to the tumor site by using a targeted beam, or a small physical source of radiation placed near or inside the tumor. Novel therapies, called radiopharmaceuticals, incorporate a small load of radiation to a molecule that preferentially enters cancer cells.
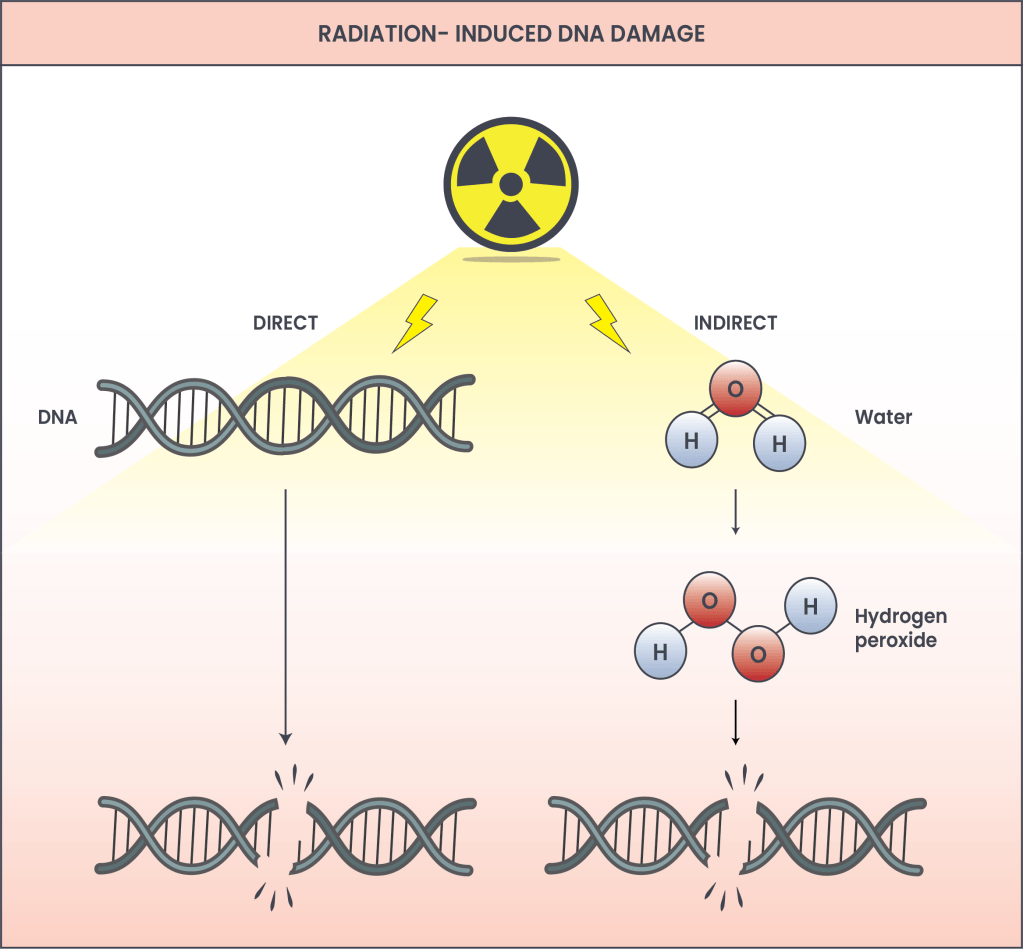
Figure 2. How radiation causes DNA damage. Radiation directly breaks DNA by adding high amounts of energy, and breaking bonds between atoms. Double-stranded DNA breaks are depicted here, however radiation causes many types of damage, including single-stranded DNA breaks and others. Indirect damage to DNA is caused by toxic chemicals produced when radiation strikes water inside of cells, generating reactive oxygen species (ROS). Hydrogen peroxide is depicted here, but others are also made. ROS can cause multiple types of DNA damage.
How healthy cells respond to radiation
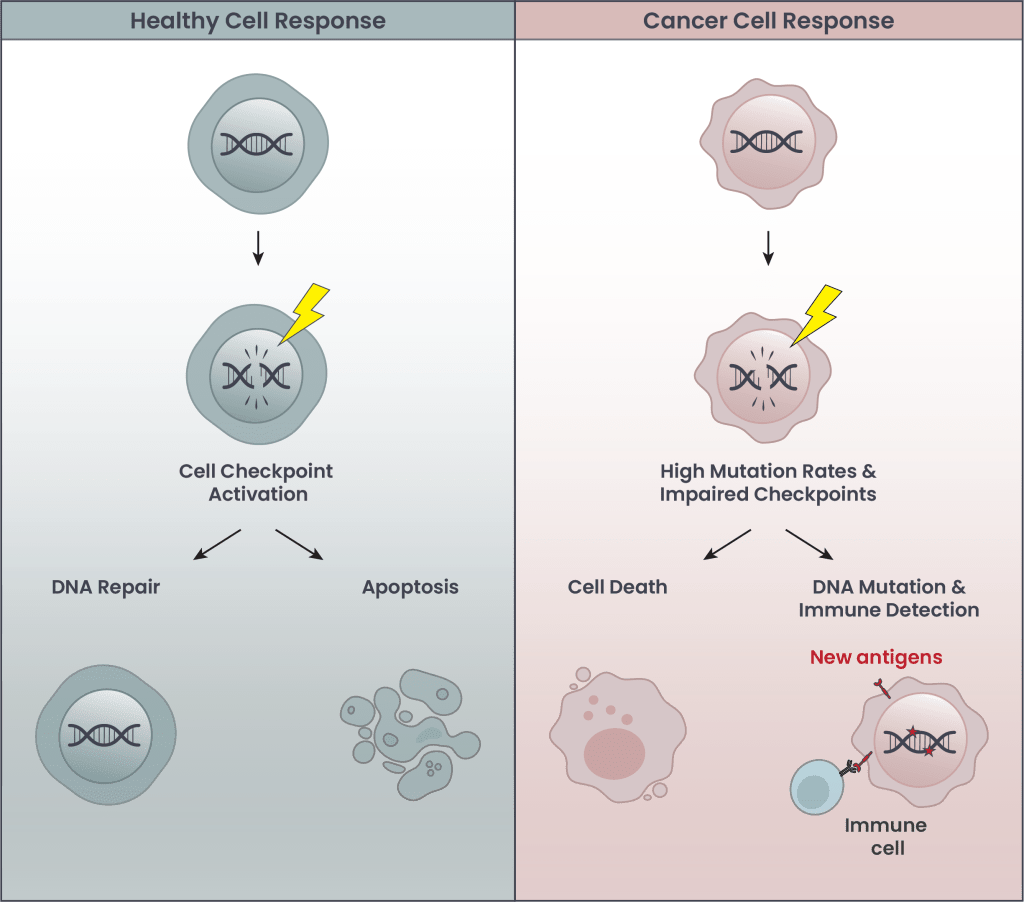
Figure 3. Responses of healthy and cancer cells to radiation. In this figure, radiation is depicted as creating DNA double-strand breaks, the most severe form of DNA damage, but it can also cause other types of DNA damage. (Left) Healthy cells with intact checkpoints respond to DNA damage by slowing growth to allow for DNA repair, or entering programmed cell death called apoptosis if damage is too extensive. (Right) Cancer cells with impaired checkpoints often leave DNA damage unrepaired, resulting in mutations that alter proteins, which are more easily detected as antigens by our immune system.
Damaged DNA presents multiple challenges to cells (Figure 3, left). One challenge is the creation of DNA mutations: when DNA gets damaged, it becomes more likely to be modified, or mutated. Since DNA is the instruction manual our cells follow to know what to do and when, avoiding mutations is important. Damaged DNA is even more likely to become mutated when cells replicate, and fast dividing cells are particularly prone to generating mutations.
Another challenge DNA damage presents is death during cell division. Cell growth is essentially cell duplication – cells grow large, double all of their DNA and other internal components, then divide into two new cells, called daughter cells. During cell division, there is an almost perfect split of components, with each daughter cell receiving identical and complete sets of DNA. DNA damage can cause strands of DNA to get stuck together, or stuck to other cell components. When bits of a cell get stuck together, but are destined for opposite daughter cells, there’s a real problem. Like siblings fighting over something fragile: it will break, and no one is going to be happy. This can result in spontaneous cell death.
To avoid these catastrophic outcomes, cells repair damaged DNA. Healthy cells even slow down their growth to facilitate accurate and complete repair in response to checkpoint activation. Checkpoints are, as their name implies, points during growth and cell division at which cells pause to check for good health. Some of the most critical checkpoints involve checking for DNA damage and pausing long enough to allow for repair. If DNA damage is too great and cannot be repaired, cell checkpoints direct cells to enact apoptosis, or other controlled cell death mechanisms, to prevent mutations from being passed on4.
How radiation targets cancer cells
Cancer cells have a modified response to DNA damage, which we take advantage of in radiotherapy. Cancer cells are driven by DNA mutations that inactivate checkpoints. This is a good example of a bug in one setting being a feature in another: DNA mutations in checkpoint proteins drive rapid cancer growth and permit the acquisition of more mutations, while avoiding cell death and growing rapidly. However, because cancer cells more often ignore DNA damage than healthy cells, toxic levels of DNA damage introduced by radiation can lead to spontaneous cell death in cancer cells. Additionally, although cancer is fueled by certain mutations, the vast majority are not beneficial, and can make the cancer look more distinct from healthy cells. Our immune system distinguishes healthy from non-healthy cells, including bacteria and cancer, to know what to target. By introducing DNA damage that goes unrepaired and results in protein mutations, which can be detected as antigens, we help our immune system distinguish cancer from healthy cells, facilitating their removal (Figure 3, right)5.
The future of radiation in cancer therapy
Although radiation was among the first cancer treatments, its uses continue to expand and improve6. The most significant area in which radiotherapy has improved is in limiting exposure of radiation to tumors, sparing surrounding healthy tissue. Proton therapy, radiation using protons, achieves this due to the properties of protons: they move through tissue doing less harm than where they stop, which can be controlled. This maximizes damage at the tumor, while minimizing damage on the path to the tumor, and completely prevents damage beyond the tumor – collectively minimizing casualties of healthy cells. To use proton therapy accurately, we must know precisely where a tumor is, and what tissue is between the radiation source and the tumor. Another innovation is the simultaneous coupling of imaging with radiation. Previously, radiation was guided based on pre-recorded images that marked tumor location and borders, potentially hitting healthy cells or missing parts of the tumor when radiation is given later on. Novel approaches entail improvements in real-time imaging to better guide radiation targeting. Thanks to incredibly dedicated and brilliant minds, we continue to push the limits of medicine to prevent one of the most heinous thieves of our body – cancer – from stealing our good health.
- Image generated by Corey Jones-Weinert (2026), using Gemini Nano Banana. ↩︎
- U.S. Nuclear Regulatory Commission (2020). Radiation Basics. Retrieved 28 March, 2026 from https://www.nrc.gov/about-nrc/radiation/health-effects/radiation-basics. ↩︎
- Borrego-Soto, G., Ortiz-López, R. & Rojas-Martínez, A. Ionizing radiation-induced DNA injury and damage detection in patients with breast cancer. Genet. Mol. Biol. 38, 420–432 (2015). ↩︎
- Yam, C. Q. X., Lim, H. H. & Surana, U. DNA damage checkpoint execution and the rules of its disengagement. Front. Cell Dev. Biol. 10, 1020643 (2022). ↩︎
- Png, S., Tadepalli, S. & Graves, E. E. Radiation-Induced Immune Responses from the Tumor Microenvironment to Systemic Immunity. Cancers 17, 3849 (2025). ↩︎
- Yan, Y. et al. Innovative approaches in precision radiation oncology: advanced imaging technologies and challenges which shape the future of radiation therapy. Front. Med. 12, 1686593 (2025). ↩︎
© 2025 WithinOncology. All rights reserved.
This article, including all text, tables, and figures, is the intellectual property of WithinOncology and its contributors. Unauthorized reproduction, distribution, or use of any content without explicit written permission is strictly prohibited. For inquiries, please contact us via the contact form.


Leave a comment