Acute Lymphoblastic Leukemia (ALL) is a fast-growing blood cancer that originates from lymphocytes1, a type of white blood cell that plays a central role in the immune system by helping the body recognize and fight infections and abnormal cells. Lymphocytes originate in the bone marrow, the spongy tissue inside bones where all blood cells, including red blood cells, platelets, and other white blood cells, are produced. After being formed, they must undergo a maturation process to become fully functional2. Some mature in the bone marrow, while others travel to the thymus or other lymphoid organs, where they develop the ability to recognize specific threats. In ALL, the bone marrow produces excessive numbers of immature lymphocytes known as lymphoblasts. These abnormal cells accumulate rapidly and crowd out healthy blood-forming cells, leading to decreased production of red blood cells, platelets, and functional white blood cells, also known as pancytopenia.
ALL is the most common type of leukemia in early childhood and accounts for approximately 25% of all childhood cancers3. In contrast, ALL is less common in adults, yet the majority of ALL-related deaths occur in the adult population4. Although children generally respond well to initial treatment and have better survival outcomes than adults, relapsed or refractory (R/R) ALL remains the leading cause of treatment failure and continues to represent a significant unmet medical need.
Homenum Revelio: Revealing the hidden cancer cells
Our immune system identifies harmful threats, including cancer cells, by recognizing surface antigens, which are molecules displayed on the outside of cells. A key component of this immune defense are T-cells, which can recognize specific antigens and destroy infected or even cancer cells. Chimeric Antigen Receptor (CAR) T-cell therapy is a form of immunotherapy that harnesses a patient’s own T-cells to recognize and eliminate cancer cells5. However, cancer cells can evade immune detection through molecular adaptations, such as expressing surface receptors that signal to the immune system not to attack. This mechanism allows cancer cells to escape immune surveillance, much like an invisibility cloak. CAR T-cell therapy is designed to overcome this evasion strategy by equipping T cells with engineered receptors that enable them to recognize and target cancer cells more effectively.
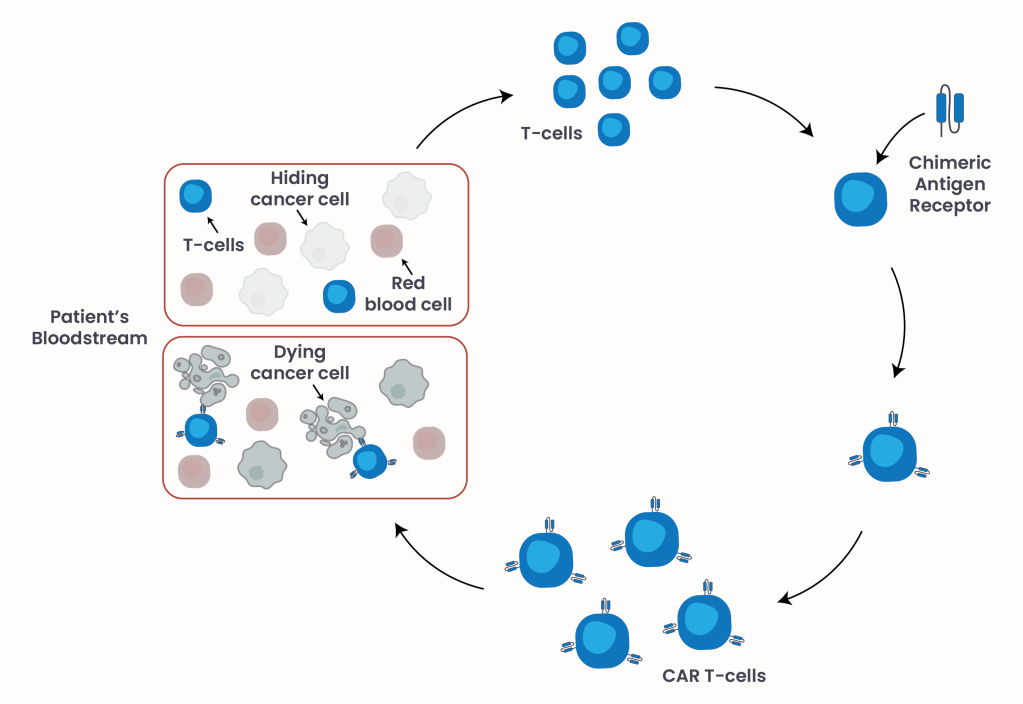
Figure 1. CAR T-cell engineering.
In this approach, T-cells are collected from the patient’s bloodstream and genetically engineered in the laboratory to express a synthetic receptor on their surface – a sort of antenna that cells use to sense their environment5. This specific receptor enables the modified T-cells to specifically recognize antigens expressed on leukemia cells. Once infused back into the patient as a one-time treatment, these engineered cells can identify and eliminate cancer cells that were previously able to evade immune detection (Figure 1).
Key elements of CAR T-cells
Over time, CAR T-cell technology has evolved to enhance three major functional properties: (1) the ability of the engineered cells to replicate, and increase in number, in the patient’s bloodstream after infusion, (2) their capacity to effectively kill cancer cells, and (3) their long-term persistence described as the ability of CAR T-cells to survive and remain functional in the patient’s body long after infusion to reduce the risk of relapse by eliminating newly emerging malignant cells.
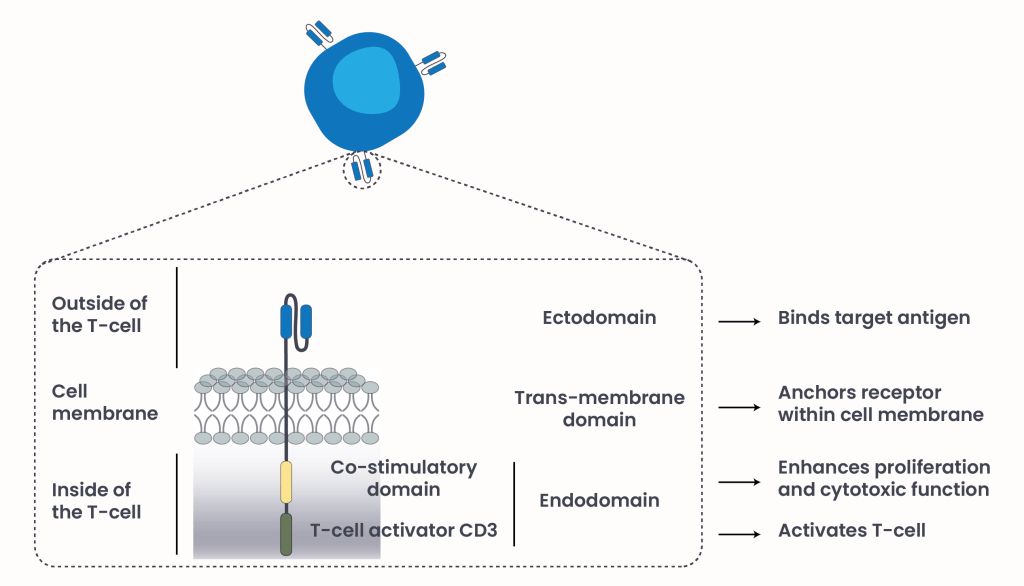
Figure 2. CAR T-cells structural components.
Currently approved CAR T-cell therapies for R/R ALL (Table 1) compose of three structural components6 (Figure 2):
- Endodomain: Located inside the T-cell, this region contains the CD3 signaling domain, which activates the T-cell once it recognizes a target antigen on a cancer cell. In simple terms, this part acts like the internal “on switch”. It also includes a co-stimulatory domain, which provides additional signals that help the T-cell multiply and stay active.
- Transmembrane domain: Anchors the receptor within the T-cell membrane, a flexible layer around T-cells separating the inside of the cell from the outside, and connects the internal signaling domain to the external antigen-binding region.
- Ectodomain: The extracellular component that specifically binds to a target antigen on cancer cells, such as CD19 or CD22.
| Kymriah (tisagenlecleucel)7 | Tecartus (brexucabtagene autoleucel)8 | Aucatzyl (obecabtagene autoleucel)9 | |
|---|---|---|---|
| Manufacturer | Novartis Pharmaceuticals Corporation | Kite Pharma Inc. | Autolus Inc. |
| FDA-approval | 2017 | 2021 | 2024 |
Targeted patient population | Children and young adults (up to age 25) with R/R ALL | Adults (18+) with R/R ALL | Adults (18+) with R/R ALL |
| Ectodomain | CD19 | CD19 | CD19 |
Table 1. Currently approved CAR T-cell therapies for R/R ALL.
The other side of the coin
The first FDA approval of CAR T-cell therapy for pediatric R/R ALL in 2017 marked a transformative milestone in oncology. It demonstrated high complete remission rates (98% of responders were MRD-negative within 3 months) and improved survival (66% of responders were alive 24 months after infusion) among ALL patients who had exhausted conventional treatments such as chemotherapy, radiation therapy, and stem cell transplantation.
However, CAR T-cell therapy also presents challenges. Treatment can be associated with serious adverse events, including cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS)10, which have been already discussed in a previous article. In addition, resistance, manufacturing time, and cost remain significant barriers.
The three primary mechanisms that contribute to CAR T-cell resistance are10:
- Antigen escape: Loss or downregulation of the target antigen, preventing CAR T-cells from recognizing leukemia cells.
- Intrinsic T-cell dysfunction: Includes T-cell exhaustion – ”tired” T-cells lose their ability to kill their targets – due to overstimulation and limited persistence in the patients’ blood stream after infusion.
- Tumor microenvironment: A complex, dynamic ecosystem surrounding the tumor comprising cancer cells, immune cells, and blood vessels along with others. TME components can have immunosuppressive activity, reducing CAR-T cell persistence and efficacy.
Beyond resistance, CAR T-cell therapy presents logistical challenges due to its individualized manufacturing process. Because the therapy must be generated from a patient’s own T-cells, production typically takes three to six weeks from cell collection to infusion. For patients with relapsed disease, this waiting period can be a critical time they may not have. In addition, CAR T-cell therapy requires complex manufacturing, coordination, and specialized facilities, creating both time delays and significant financial burdens for patients and healthcare systems. In contrast, conventional therapies such as oral medications are available off the shelf and can be administered immediately. However, while CAR T-cell therapy is primarily used in relapsed or refractory settings, its high efficacy has prompted discussions about earlier use in treatment sequences to potentially prevent relapse.
Future direction for novel strategies
Ongoing research is focused on improving the efficacy, durability, and safety of CAR T-cell therapy through several innovative strategies (10). These include (1) the development of multi-targeting constructs, such as dual CD19/CD22 CAR T-cells, to reduce the risk of antigen escape; (2) further engineering improvements on enhancing persistence and long-term functional activity; (3) combination strategies with other immunotherapies to strengthen anti-tumor responses; and (4) improved patient stratification through advanced diagnostic tools and screening technologies to enable more precise categorization based on specific patient characteristics, helping identify those most likely to benefit from treatment.
Overall, CD19-targeting CAR T-cell therapy represents a major breakthrough in the treatment of R/R ALL and improves patient outcomes greatly. Although challenges such as toxicity, resistance, and manufacturing complexity remain, continued scientific innovation is steadily refining and expanding the therapeutic potential of this transformative approach.
- National Comprehensive Cancer Network. (Version 2025), Guidelines for patients. https://www.nccn.org/home. Retrieved February 27 2026 from https://www.nccn.org/patientresources/patient-resources/guidelines-for-patients. ↩︎
- Gómez-Manríquez, J. et al. B cell development: transcriptional regulation and immunological mechanisms in homeostasis. Front. Immunol. 16, 1593338 (2025). ↩︎
- American Cancer Society (22 Jul. 2025), Key Statistics for Acute Lymphocytic Leukemia (ALL). https://www.cancer.org. Retrieved February 27 2026 from https://www.cancer.org/cancer/types/leukemia-in-children/key-statistics.html. ↩︎
- American Cancer Society (22 Jul. 2025), Key Statistics for Childhood Leukemia. https://www.cancer.org. Retrieved February 27 2026 from https://www.cancer.org/cancer/types/acute-lymphocytic-leukemia/about/key-statistics.html. ↩︎
- Shamim H, Jhakri K, Al-Shudifat M, Sumra B, Kocherry C, Malasevskaia I. Chimeric Antigen Receptor T-cell Therapy in the Treatment of Pediatric Acute Lymphoblastic Leukemia: Efficacy, Safety, and Future Directions. Cureus. 2025 Jul 31;17(7):e89172. doi: 10.7759/cureus.89172. PMID: 40895845; PMCID: PMC12398623. ↩︎
- Zugasti I, Espinosa-Aroca L, Fidyt K, Mulens-Arias V, Diaz-Beya M, Juan M, Urbano-Ispizua Á, Esteve J, Velasco-Hernandez T, Menéndez P. CAR-T cell therapy for cancer: current challenges and future directions. Signal Transduct Target Ther. 2025 Jul 4;10(1):210. doi: 10.1038/s41392-025-02269-w. PMID: 40610404; PMCID: PMC12229403. ↩︎
- Novartis Pharmaceuticals Corporation (Jan. 2025), I am KYMRIAH. https://www.novartis.com/us-en/about. Retrieved February 27 2026 from https://www.kymriah-hcp.com/diffuse-large-b-cell-lymphoma-adults. ↩︎
- Kite Pharma Inc. (Aug. 2025), Redefine their storyline. https://www.kitepharma.com/. Retrieved February 27 2026 from https://www.tecartushcp.com/car-t-cell-therapy/acute-lymphoblastic-leukemia. ↩︎
- Autolus Inc. (Jan. 2026), Elevate CAR T-cell therapy with AUCATZYL. https://www.autolus.com. Retrieved February 27 2026 from https://www.aucatzylhcp.com/ ↩︎
- Li F, Zheng L. Precision immunotherapy with CAR-T cells in pediatric B-cell acute lymphoblastic leukemia: advances and unanswered challenges. Front Oncol. 2026 Jan 14;15:1691189. doi: 10.3389/fonc.2025.1691189. PMID: 41613540; PMCID: PMC12846953. ↩︎
© 2025 WithinOncology. All rights reserved.
This article, including all text, tables, and figures, is the intellectual property of WithinOncology and its contributors. Unauthorized reproduction, distribution, or use of any content without explicit written permission is strictly prohibited. For inquiries, please contact us via the contact form.


Leave a comment